

Empowering Healthcare Worldwide

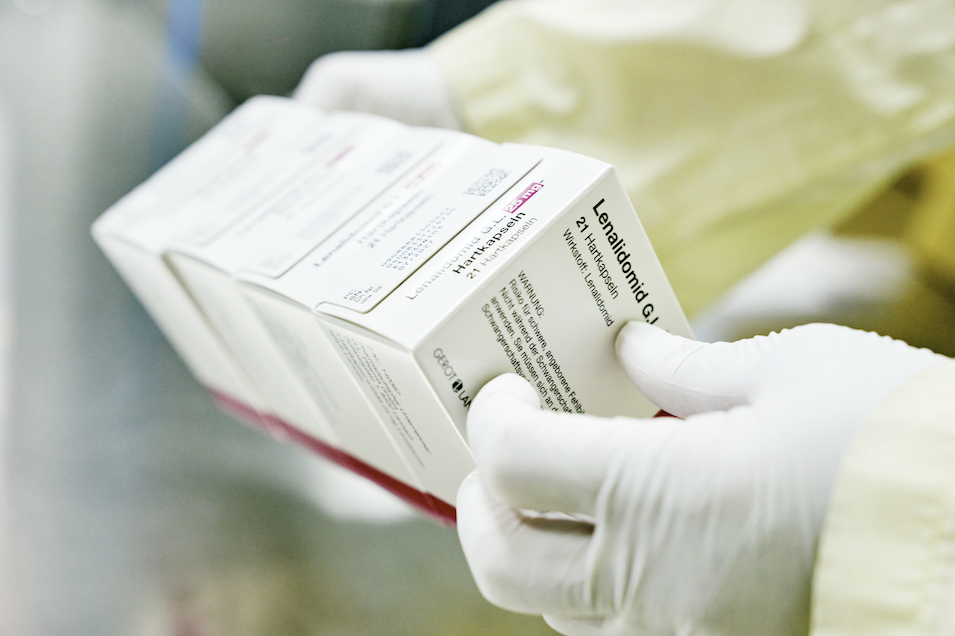

Differentiated Portfolio
We market over 250 commercial products across 90+ markets globally, with pipeline supported by our hybrid R&D+BD strategy. With expertise in high‑barrier oncology and complex generics, we focus on addressing unmet medical needs. Through targeted acquisitions and licensing of branded medicines, 505(b)(2) products, biosimilars, and NCEs, we continue to build a differentiated portfolio that bolsters treatment access for patients worldwide.


Generics
Therapeutic Areas
With deep R&D expertise and proven regulatory strength, we deliver differentiated therapies across multiple disease areas, with a strategic focus on the fast-evolving fields of oncology and immunology.
We strategically invest in developing and commercializing a differentiated portfolio of niche, high-complexity pharmaceuticals. Leveraging our experienced R&D team, advanced research and manufacturing capabilities, we drive a robust pipeline designed to address medical needs and remain at the frontiers of medical innovation.


Drug Risk Management Plan


CRO Services
Norwich Clinical Services offers comprehensive clinical research solutions to the pharmaceutical and biotech industries, specializing in Bioavailability/Bioequivalence Studies, Clinical Trials, Pharmacovigilance, Bioanalytical Services, and Training. Our expert team based in Bangalore, India, ensures precision and efficiency in delivering reliable results.
Our unique combination of clinical expertise, therapeutic experience, equipment and processes makes us able to manage and overcome challenges with specific focus on maximizing project budgets and meeting timelines.


